DIRECTIONS
Read each question carefully. For a multiple-choice question, determine the best answer to the question from the four answer choices provided. For a griddable question, determine the best answer to the question. Then fill in the answer on your answer document.
1 An atom is shown below.
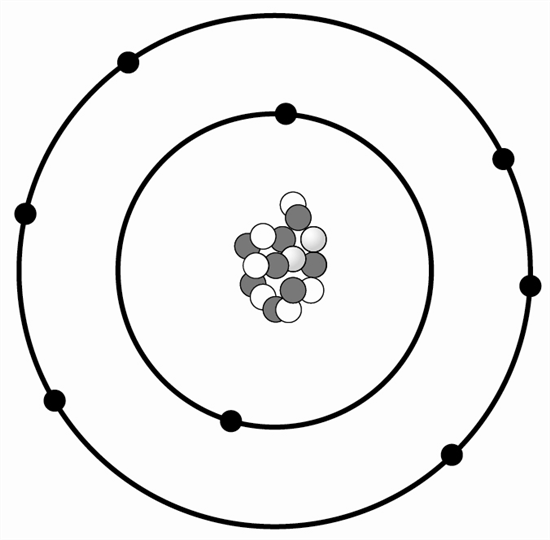
Which statement below best describes where subatomic particles are located in the atom?
A Electrons are in the nucleus and have a negative charge.
B Protons and neutrons are in the nucleus and both are positively charged.
C Neutrons are in the nucleus and provide no mass to the atom.
D Protons are in the nucleus and provide a positive charge to the atom.
2 Becky was trying push a box to the other side of her bedroom. Her sisters, Janice and Alex, decided to help her.
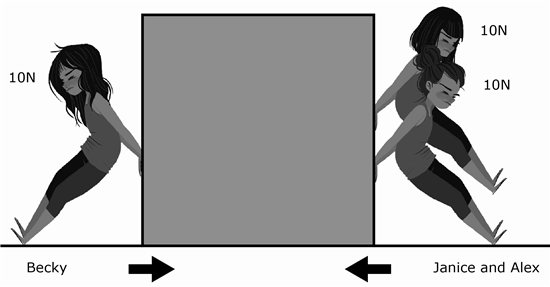
Are Janice and Alex helping Becky?
F Yes, because Janice and Alex's added forces will help push the box further faster.
G No, because Becky's force will push the box further faster towards Janice and Alex's forces.
H Yes, because Janice and Alex's forces cancels out the mass of the box.
J No, because Janice and Alex's added forces is pushing against Becky's force which will push it towards Becky.
3 The element with 9 protons, 10 neutrons, and 9 electrons is �
A boron
B neon
C beryllium
D flourine
4 GPS (Global Positioning System) devices installed in modern cars can make navigation in unfamiliar areas much smoother.
Suppose a GPS device instructs a driver to travel at 55 kilometers per hour north. This instruction is an example of...
F acceleration
G speed
H position
J velocity
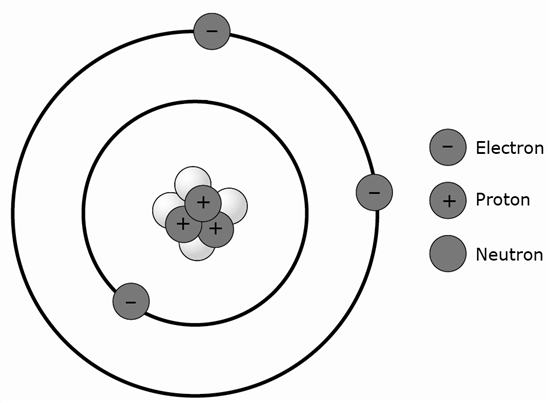
5 An atom of which element is represented by this model?
A Hydrogen (H)
B Lithium (Li)
C Bromine (Br)
D Radon (Rn)
6
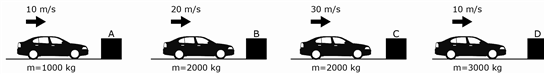
In each of the above situations, an identical car is heading towards a solid wall. Which of the following choices correctly ranks the magnitude of the force of the collision from greatest to least?
F A, D, B, C
G D, C, B, A
H C, B, A, D
J C, B, D, A
7 The model of The Periodic Table below shows the locations of four elements.
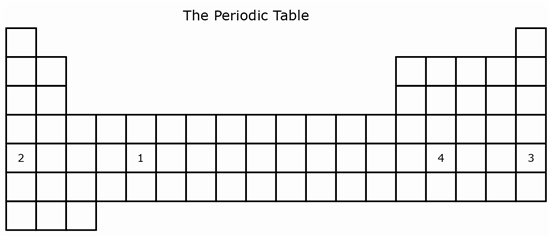
A student is asked to identify the metal in the fifth period that is most reactive. At which location in this periodic table would the element most likely be found?
A 1
B 2
C 3
D 4
8 A 1 kilogram ball is rolled along three tracks shown below. In all three instances, a resting ball is pushed with equal force from point X in the direction indicated.
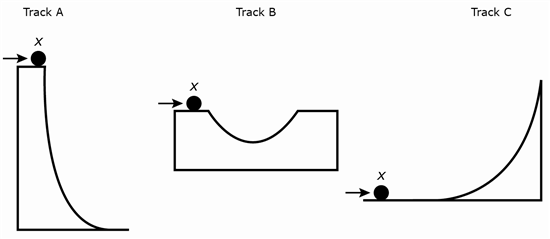
Which of the track configurations would cause the greatest change in speed?
F Track A would have the greatest change in speed.
G Track B would have the greatest change in speed.
H Track C would have the greatest change in speed.
J All of the tracks have the same change in speed.
9 A carbon (C) atom has 6 electrons orbiting a nucleus. The nucleus is made up of 6 protons and 6 neutrons. What is the total net charge of this carbon atom?
A 6
B 12
C 0
D 18
10 A graph is shown below.
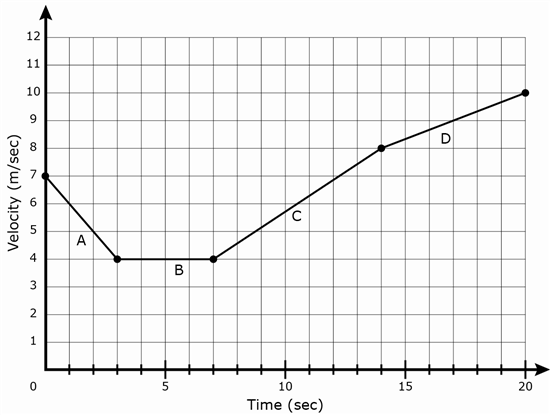
Which letter represents the segment on the graph where an object is not accelerating?
F A
G B
H C
J D
11 Jan wants to experiment with an element that reacts like chlorine (Cl), but has a smaller atomic mass. Which element should she select for the experiment?
A F
B Br
C Ar
D S
12 Four grocery clerks pushed carts full of different produce across the parking lot. Each clerk pushed with the same amount of force. Which cart had the greatest change in speed?
F 
10 pineapples
m = 10 kg
H 
30 oranges
m = 8.5 kg
G 
6 watermelons
m = 20 kg
J 
50 bananas
m = 9.15 kg
13 It takes a car 3.6 seconds to get from 0 m/s to 25 m/s. This is an example of �
A constant speed
B velocity
C acceleration
D inertia
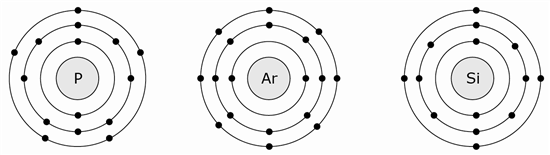
14 What is true about all three models?
F All three atoms have equal reactivity.
G All three atoms are in the same period.
H All three atoms are very heavy.
J All three atoms are in the same group.
15 A car is shown below.
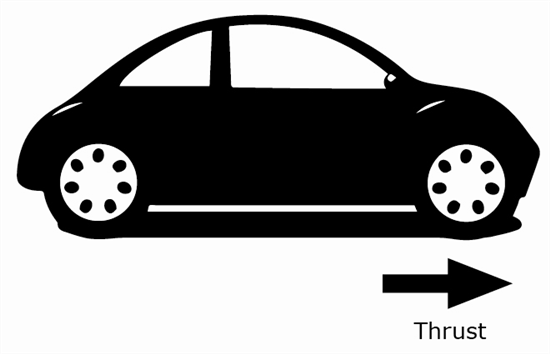
What acceleration is necessary to move a car forward with a mass of 150.0 kg and a net force of 3,750 N?
Record your answer and fill in the bubbles on your answer document. Be sure to use correct place value.
16 A piece of Germanium is shown below.

The metalloid Germanium (Ge) is similar to a metal because it �
F is a good conductor of heat and electricity
G is a poor conductor of heat and electricity
H does not usually have much luster
J is usually brittle
17 A Chlorine (Cl) atom is shown below.
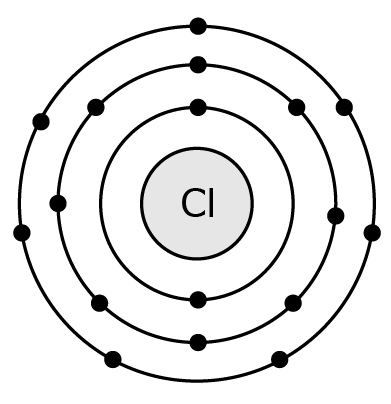
An atom of Chlorine (Cl) has an atomic number of 17 and a mass of 35, how many neutrons does it have?
Record your answer and fill in the bubbles on your answer document. Be sure to use the correct place value.
18 A train trip from New York City to Boston, MA takes 120 minutes. The distance between New York City and Boston is 350 km. What is the average speed of the trip from NYC to Boston in kph?
F 30 kph
G 70 kph
H 175 kph
J 100 kph
19 Based on what you know about atomic structure, silicon (Si) has a very similar reactivity to �
A 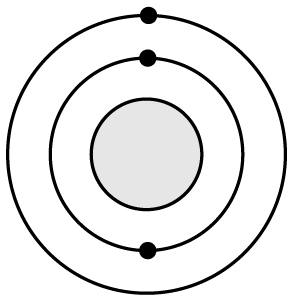
B 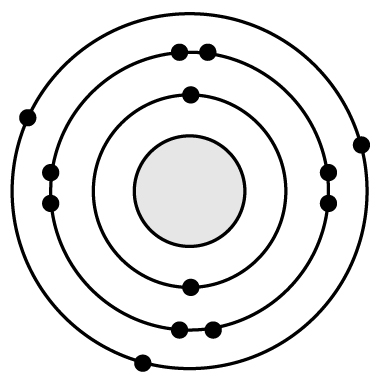
C 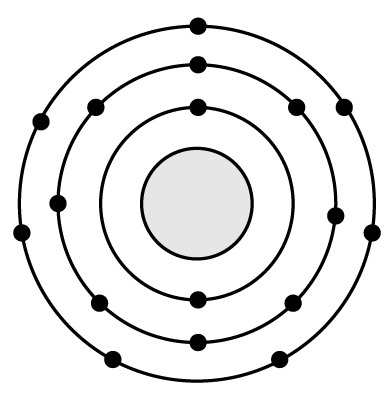
D 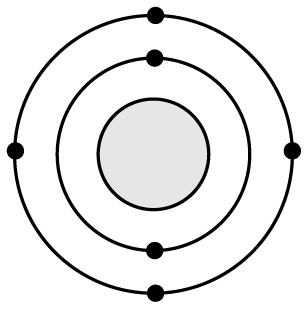
20 Which of the following compounds is organic?
F NaCl
G
H O
H
CH N O
J Fe O

DIRECTIONS
Answer the following question in the box labeled "Short Answer 1" on your answer document using complete sentences.
Immediately following an earthquake, some large buildings may continue to rock for a period of time. More massive buildings continue to move for longer than smaller buildings. Which Newton's law does this demonstrate? Define the law and justify why this law best explains what is going on in the example.



















